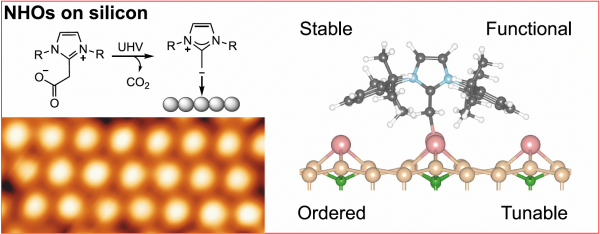
N-Heterocyclic olefins (NHOs) are carbon-based organic ligands closely related to N-heterocyclic carbenes (NHCs). NHCs are well established moieties in organometallic chemistry and homogeneous catalysis and have attracted much interest recently due to their ability to form self-assembled monolayers (SAMs) on transition metal surfaces and even on silicon (see CNR-ISM press release), thus offering an alternative to thiol-based self-assembly on metals as well as a new paradigm for forming controlled organic/silicon interfaces.
In NHOs, an alkylidene unit (CH2 group) is formally attached to the NHC.
The additional electron-rich double bond is described by a resonance between the neutral olefin and a zwitterionic ylide mesomer, offering strong σ-donation ligand possibilities. However, NHOs have until now not been used in surface modification, despite possessing the same flexibility of NHCs offered by side-group and backbond modification.
In this study, the German groups synthesised and deposited two kinds of NHO on a passivated silicon surface. STM and XPS measurements revealed the formation of ordered monolayers with high thermal stability and work function modification.
Theoretical calculations by the CNR-ISM and UTV group proved that the NHOs adsorb in ylide, and not olefinic, form, and revealed the presence of different upright or flat binding modes depending on the presence of bulky side groups, thus offering a further tuning parameter over the chemical properties of the SAMs.
The study opens exciting new prospectives in surface functionalization.
For info: Conor Hogan

 English (UK)
English (UK)  Italiano (Italia)
Italiano (Italia)