
UV-visibile Spectrophotometer Perkin Elmer Lambda 950
ATR - FT-IR Spectrophotometer Shimadzu Prestige 21
Elvira Maria Bauer -
TECHNICAL SPECIFICATIONS
-
UV-Vis: (PerkinElmer, Lambda 950)
- Spectral range 190 – 860 nm, resolution from 5 nm up to 0.05 nm; high performance PM detector in the UV-Vis range
- UV lamp – deuterium, Vis lamp – tungsten halogen, flexible exchange Double beam, double monocromator Dual sample compartment, permits easy and quick sample module exchange (trasmittance/absorbance, integrating sphere, URA) Optical bench custumaziable Two cuvette holders sample and reference)
-
FT-IR: (Shimadzu, Prestige 21)
- spectral range 7,800 – 350 cm-1, resolution up to 0.5 cm-1, S/N ratio 40,000 : 1
- ceramic light source He-Ne laser DLATGS detector KBr/Au beam splitter Dessicated Michelson interferometer Single beam Automatic accessory detection Sample holder for NaCl, KBr and CsI discs ATR basement (Specac Golden Gate)
AVAILABLE TECHNIQUES
-
Spettroscopia UV-Vis
- Quantitative and qualitative transmittance/absorbance measurements of liquids and transparent thin film, determination of extinction coefficients, reaction cinetics
- Wavelength and energy scan, fixed wavelength scan and cinetics
- Quartz cuvettes with 1 mm and 10 mm optical path
- Film sample holder (3 x 8 mm)
- Centrifuge and ultrasonic bath for sample preparation
-
FT-IR spectroscopy
- Qualitative transmittance/absorbance measurements on solid and liquid organic, hybrid inorganic-organic and inorganic materials
- 13 mm and 25 mm dyes (Specac)
- ATR (Attenuated Total Reflectance) accessory (Specac Golden Gate) with diamond crystal base plate; sapphire, liquid and flexible material anvil; manual pressure control by micrometric screw; for analysis of pure solids, liquids and flexible films
- NaCl and CsI discs Thermostatic oven for KBr desiccation
SAMPLE
UV-Vis:
-
Liquids or solids dissolved in low absorbing solvents (2-5 ml), solutions must be clear, quantities depend on the extinction coefficient of the investigated material
FT-IR:
-
Powders (2-5 mg) of organic, hybrid or inorganig materials
Liquids (1-2 ml), only ATR
- Flexible films and plastics of min. 3 x 5 mm up to max. 10 x 10 cm size
- ATR accessory cannot be used for diamond-based or highly corrosive materials
USE FOR
- Chemical and Pharmaceutical Industry
- Catalysis
- Cleaning and water purification.
- Quality control
CASE STUDIES
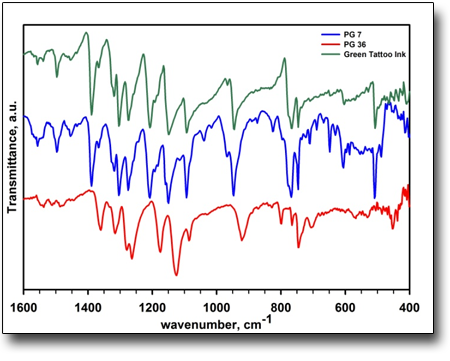
Determination of the colouring component in commercial tattoo inks..
FT-IR spectroscopy is one of the most important tools to determine the nature of functional groups in a wide range of materials. The present example regards the analysis of several green tattoo inks from a well-known american producer by spectrophotometric and microscopic techniques. Registration of FT-IR spectra evidenced the presence of a green phthalocyanine based pigment which resulted to be different from the one declared on the label of the ink bottle and the accompanying SDS card. All investigated green inks showed the presence of Pigment Green 7 (PG 7, hexadecachloro copper phthalocyanine) which is not allowed in tattoo ink preparations in Switzerland and some European countries instead of the declared and permitted Pigment Green 36 (PG 36, hexachloro decabromo copper phthalocyanine). Furthermore FT-IR spectroscopy associated with Raman spectroscopy ascertained the presence of only one yellow monoazo pigment (PY 14) in some green ink samples although if otherwise declared on the labe.
See: E.M. Bauer, T. De Caro, P. Tagliatesta, M. Carbone, Dyes&Pigments 167, 225 (2019)
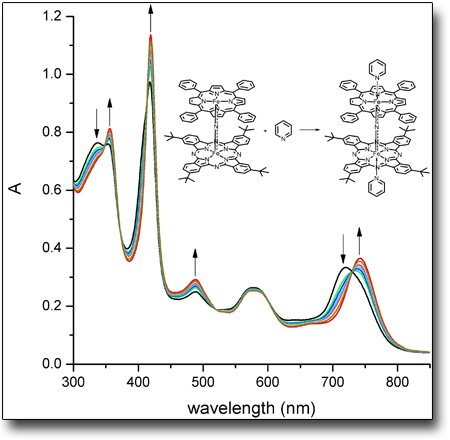
Kinetic studies by UV-Vis spectroscopy.
UV-Vis spectroscopy allows to study the kinetic behavior of several chemical reactions by measuring the spectral change related to variations in the concentrations of reactants and/or products over time. In the reported example, the absorption spectrum of a -nitrido phthalocyanine-porphyrin bridged system in dichloromethane is modified by reaction with a given amount of pyridine: the progressive complexation of the two central metals by the pyridinic nitrogen changes the spectrum shape to that of the final product. The analysis of the spectra recorded over time allows to determinate the reaction rates and the related kinetic constants.

 English (UK)
English (UK)  Italiano (Italia)
Italiano (Italia)